Current Projects
Geroscience Computational Core
To make CUIMC a global leader in geroscience by integrating cutting-edge aging biomarkers into every stage of the transnational research pipeline.
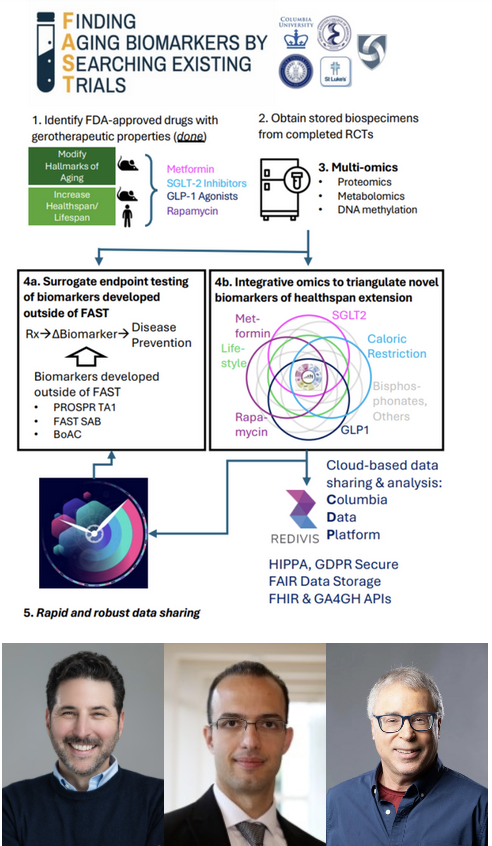
FAST Initiative
Aging is the leading cause of most chronic diseases driving population burden of disability and premature mortality. The decline our bodies experience as we grow older is accepted as normal. It shouldn’t be. We can see around us the examples of successful aging; people live through their 80s and 90s cognitively intact and in good health. Breakthroughs in geroscience establish the biology of aging can be modified; interventions in laboratory animals that slow biological processes of aging increase healthspan. These preclinical discoveries suggest that, for humans, maintaining health and function into the 9th decade of life and beyond is possible for all of us. FAST is motivated by the long-term goal of enabling more people to realize this potential for healthspan extension. To achieve this goal, we aim to move biomedical research away from geriatrics-clinic whack-a-mole, in which we develop treatments for symptomatic diseases one at a time, and toward therapies that maintain health and prevent disease. Preclinical studies are bringing these therapies within reach. But translating them in human trials faces the challenge that, unlike in short-lived laboratory models, determining impacts on human healthspan will require decades of follow-up. To overcome this challenge, biomarkers are needed that are responsive to intervention over timescales of months-to-years and which can provide surrogates for the long-term impacts of novel therapies on healthspan. The immediate objective of FAST-PROSPR is to establish sensitive and specific biomarkers that can be tested to determine whether an intervention is slowing the biology of aging and extending healthspan (AY2AX000158 - ARPA-H PROSPR).
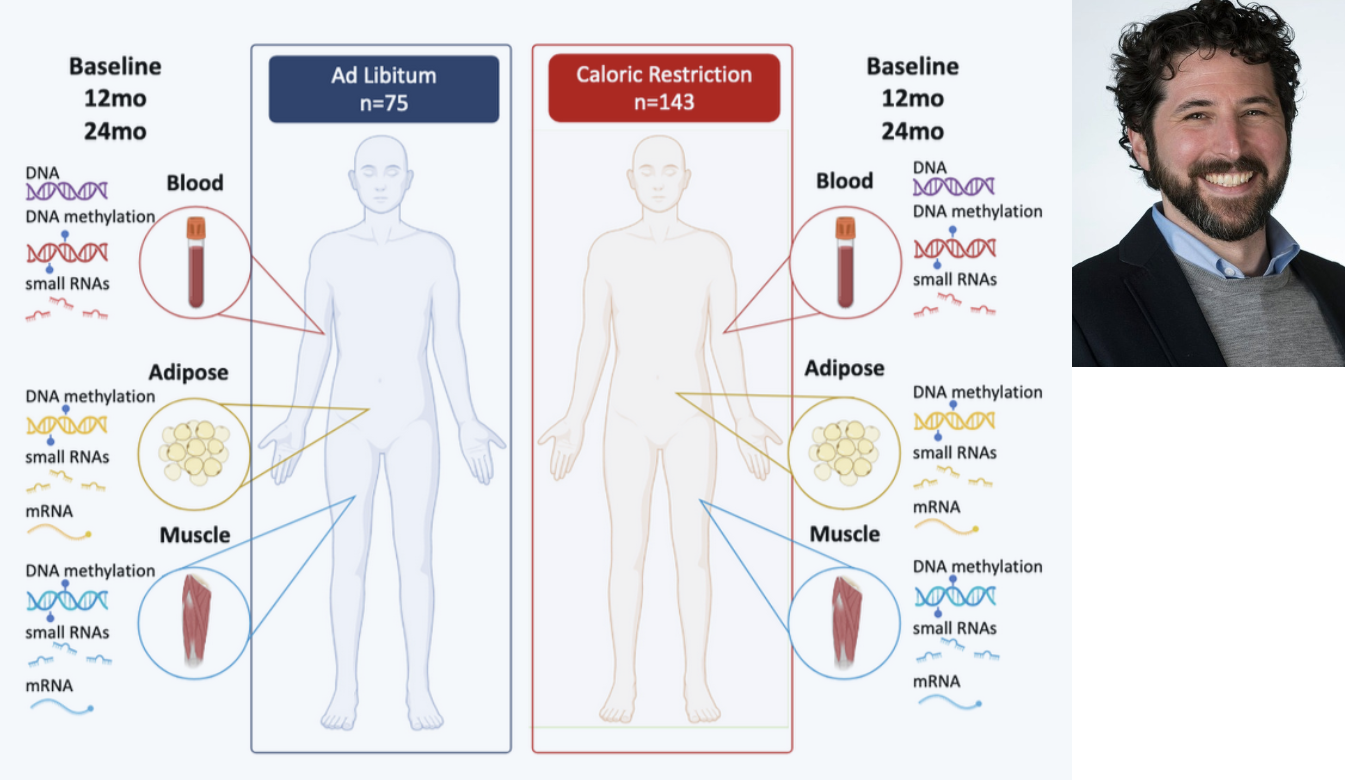
The CALERIE Genomic Data Resource
CALERIE is the first-ever randomized controlled trial of caloric restriction in healthy, non-obese adults (NCT00427193). We are conducting multi-omics analysis of tissues collected during the original CALERIE trial as well as a 10y post-intervention follow-up study (LEGACY, NCT05651620) We are studying how caloric restriction, which is proven to increase healthy lifespan in many animal models, may affect biological aging in humans, and how these effects may persist over a decade after conclusion of the intervention trial. (National Institute on Aging R01AG061378, 2R01AG061378, 2R33AG070455)
Genetic Analysis of the Dutch Hunger Winter Families Study to Boost Rigor and Robustness for Testing In-Utero Famine Effects on Aging-Related Health Conditions and Biological Aging.
The graying global population makes interventions to extend healthy lifespan a public heath priority. Health insults during the perinatal period are linked with risk for aging-related health conditions, including obesity, type 2 diabetes, and cardio-metabolic disease. If these associations are causal, interventions to prevent perinatal insults and to reverse their biological damage could delay disease onset and prolong healthspan, the length of time a person remains healthy. (National Institute on Aging R01AG066887)
The MyGoals for Healthy Aging Multi-Center Randomized Controlled Trial
MyGoals for Healthy Aging is a randomized controlled trial testing if a novel anti-poverty program slows biological aging in highly disadvantaged public housing recipients, including by inducing changes in risk factors for Alzheimer's disease/Alzheimer's disease-related dementias. (National Institute on Aging R01AG073402)
Testing Effects of Cash Transfers on Biological Aging and Risk for Alzheimer's Disease
The primary objectives of this project are to assess the effects of a cash transfer program on the prevention of biological aging and dementia risk in younger and older individuals. Additionally, we aim to investigate the association between biological aging and cognitive function in this population. The results of this research could hold substantial implications for public health by revealing the causal and long-term health consequences of socioeconomic interventions on health. Moreover, it will offer fresh perspectives on the levels of biological aging and Alzheimer’s Disease risk within the context of lower- and middle-income countries. (National Institute on Aging R01AG087158)
Life-course Sociogenomic Analysis of Social Inequalities in Aging.
Life-course social science links early-life social disadvantage with adverse outcomes in aging. A gap in knowledge is how social disadvantage is biologically embedded, leading to social inequalities. A hypothesis is that social disadvantage actually hastens aging. While everyone ages chronologically at the same rate, biological changes with aging may proceed faster for some than others. These changes are thought to be a root cause of disease/disability and an intervention target to extend healthy lifespan. (Russel Sage Foundation BioSS Grant 1810-08987)
Developing Epigenetic Measurements
to Forecast Long-Term Benefits of Early-Life Interventions.
Early-life adversity a major driver of health and social problems across the life-course. Several interventions, including home-visitor programs, show potential to protect young children from lifelong damage arising from early-adversity. These interventions increase parental nurturing and prevent some early-life stress exposures. In the short- to medium-term, they benefit families and children. But longer-term outcomes vary substantially across children. (Jacobs Foundation Young Scholars Award; CIFAR CF-0249-CP22-034)